 🖨️ Print post
🖨️ Print post
Article Summary
• Successful traditional diets provided many nutrients that cooperate with one another to produce excellent health. This article provides several illustrative examples of this type of cooperation.
• Methionine from muscle meat contributes to cell growth and repair, cellular communication, antioxidant defense, and detoxification. In order to fulfill these functions, however, methionine must be balanced with B vitamins, choline, and glycine from organ meats, egg yolks, legumes, leafy greens, skin and bones.
• Vitamins A, D and K cooperate to protect our soft tissues from calcification, to nourish our bones and teeth, and to provide children with adequate growth. We obtain these nutrients together by consuming organ meats, cod liver oil, fatty fish, grass-fed animal fats, green and orange vegetables, and fermented plant foods.
• Magnesium is required for every process in the body. Among its many interactions, magnesium is required for proper calcium metabolism. Magnesium is abundant in many plant foods and some seafood, but there is little magnesium in meat and almost none in refined sugar and refined grains. Consuming a balanced diet devoid of refined sugar and refined grains is the best way to obtain adequate magnesium.
• These interactions demonstrate that biology is very complex. Rather than thinking about whether certain nutrients from traditional diets are good for us or bad for us, we should seek to understand how they all work together in proper balance to promote radiant and vibrant health.
Synergy and Context with Dietary Nutrients
Successful traditional diets provided a rich array of nutrients that cooperated with one another to produce vibrant health. As modern diets have shifted towards nutrient-poor foods fortified with the favored nutrients du jour, we have gazed askance at the degeneration that has resulted and embarked on a series of searches for the dietary villains that we imagine lurking in the shadows.
We have blamed heart disease on cholesterol, mortality on meat, osteoporosis on vitamin A, and diabetes on fat. Yet somehow it has eluded us that we are asking all the wrong questions. Biology is not a war between good molecules and evil molecules, nor is it a war between “wholesome” natural foods like vegetables and “poisonous” natural foods like meat. Biology is a system wherein many parts work together in synergy to produce a context within which each part benefits the whole. Several examples of this type of synergy follow.
METHIONINE, B VITAMINS, GLYCINE
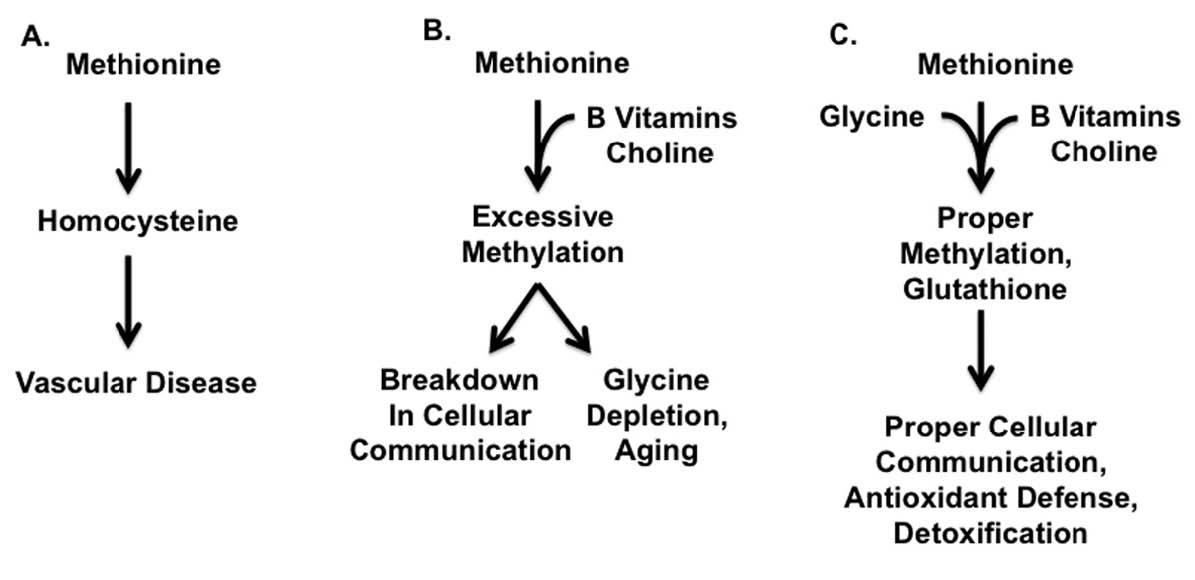
Successful traditional diets provided muscle meats together with organ meats and gelatinous materials such as bones, gristle and other connective tissue. These combinations provided a healthy balance between the methionine found in muscle meats, the B vitamins found in organ meats, and the glycine found in connective tissue. Modern diets, by contrast, provide abundant quantities of methionine-rich muscle meats while organs and connective tissue have fallen by the wayside. The result of this imbalance is that methionine is unable to fulfill its proper cellular functions and generates toxic byproducts instead, while the supply of glycine is depleted. Together, these changes are likely to contribute to reduced longevity and chronic disease (Figure 1).
Methionine is an amino acid that we obtain from most dietary proteins, but is especially abundant in animal proteins (Table 1). As shown in Figure 2,1-3 folate and vitamin B12, and to a lesser extent vitamin B6, niacin, and riboflavin, assist methionine in carrying out one of its major cellular functions: the addition of a single carbon atom together with a small assortment of hydrogen atoms to a wide variety of molecules, a process known as “methylation.” Methylation is important for the synthesis of many cellular components and for the regulation of gene expression. As a result, it is critical for the maintenance and repair of existing tissue, the building up of new tissue, and cellular communication. Methylation is especially important for the passing along of epigenetic information from parent cells to their daughter cells as they multiply. Liver is rich in all of the B vitamins important to this process. Muscle meats provide smaller amounts of most of them, but are relatively poor in folate. Folate is found primarily in liver and legumes, with modest amounts in egg yolks and some seeds, seafood, and leafy greens (Table 2). When any of these vitamins is missing, methionine fails to contribute properly to methylation and instead generates homocysteine, a potentially toxic byproduct that may contribute to cardiovascular disease.4
In support of the relevance of these pathways to human nutrition, a randomized, placebocontrolled trial showed that three months of combined supplementation with folic acid and vitamin B12 lowered homocysteine concentrations.5 In the same study, a single large dose of methionine temporarily increased homocysteine concentrations, while supplementation with B vitamins protected against this effect. This study demonstrates the critical need for balance between methionine and these B vitamins, and suggests that many people may not be getting enough folate or vitamin B12 to properly handle the methionine they are obtaining from muscle meats.
As shown in Figure 2,1-3 once our needs for methylation are met, we use vitamin B6 and glycine to convert any additional methionine in our diet to glutathione, which is the master antioxidant and detoxifier of the cell as well as a key regulator of protein function. The conversion of methionine to glutathione is not instantaneous, however, and our liver requires a buffer system to protect itself against excessive methylation and the accumulation of homocysteine.
This buffer system is comprised primarily of three nutrients: glycine, which is found most abundantly in bones and other connective tissue (Table 3); choline, which is found primarily in liver and egg yolks (Table 4); and betaine, which we can either make within our own bodies from choline or obtain directly in our diets from spinach, wheat, and beets (Table 5). Muscle meat provides its own vitamin B6, but provides relatively little glycine, choline and betaine. In order to safely use extra methionine from muscle meat to support our antioxidant defenses and detoxification systems, we therefore must balance muscle meat with liver and egg yolks as well as with soups, gravies, sauces, or other creative dishes made from bones and other connective tissue, including skin. As useful adjuncts to these foods, some people may also benefit from incorporating spinach, wheat or beets into their diet.
Several studies support the relevance of these pathways to human nutrition. In one such study, a large dose of methionine increased the excretion of a metabolic byproduct of glycine, choline and betaine in the urine,6 suggesting that excess methionine causes the irreversible loss of these nutrients. Randomized, placebo-controlled trials have shown that two weeks’ supplementation with choline7 or six weeks’ supplementation with betaine7 lowered homocysteine levels both in the fasting state and after consuming a large dose of methionine. In a similar study, three months’ supplementation with vitamin B6 made a small improvement in homocysteine levels after a large dose of methionine.5
There are, unfortunately, very few nutritional studies using glycine because scientists have not considered it an “essential” amino acid. Although our bodies can synthesize glycine, primarily from the amino acid serine, one group of scientists recently estimated that our ability to produce glycine may fall short of our needs for this amino acid by up to ten grams per day.1 This is roughly the equivalent of an ounce of bone meal each day. These authors pointed out that markers of glycine deficiency appear in the urine of vegetarians, people consuming low-protein diets, children recovering from malnourishment, and pregnant women. They further suggested that most of us adapt to a subtler degree of glycine deficiency by decreasing our own turnover of collagen, which may lead to the accumulation of damaged collagen with age, thereby contributing to arthritis, poor-quality skin, and many of the other negative consequences of aging. Indeed, while some studies have shown that restricting dietary methionine lengthens the lifespan of rats and while these have generated a great deal of interest, a similar study recently showed that the same effect can be achieved by supplementing the diet with extra glycine.8
Some authors have recently suggested that a vegan diet would lengthen lifespan because of its naturally low methionine content.9 If methionine restriction primarily increases lifespan by increasing the ratio of glycine to methionine, however, then this suggestion could not be more wrong, because vegetarians show signs of glycine deficiency.1 Vegan diets are low in total biologically available protein, not just methionine. Human studies suggest that low-protein diets waste glycine by using it simply as a source of much-needed nitrogen.10
A better way to improve the balance of glycine to methionine would be to replace a substantial proportion of muscle meats in the diet with bones and skin. Adding organ meats, egg yolks, and plant foods rich in folate and betaine to the diet would also be likely to improve longevity by working with glycine to support the safe and effective utilization of methionine. When these nutrients are all provided in rich supply, methionine supports the growth and repair of tissues, our defense against oxidants, detoxification and proper cellular communication.
VITAMINS A, D AND K2
Successful traditional diets also provided a balance between vitamins A, D, and K2. Vitamin A is most abundant in liver and fish liver oils, such as cod liver oil.11 Plant foods rich in carotenoids also provide vitamin A, although they do so much less reliably than liver and cod liver oil because the ability to convert carotenoids to vitamin A varies about ten-fold between individuals. 12
Vitamin D is most abundant in cod liver oil and fatty fish. Sunshine is also an important source of vitamin D, though our ability to use sunshine to synthesize this vitamin depends on where we live, our skin color, how much time we spend outdoors, and the type of clothing we wear.13
Vitamin K2 is found primarily in animal fats and fermented foods.14 We can also synthesize vitamin K2 from the vitamin K1 found in leafy green vegetables, but this conversion seems to be very inefficient in humans. To a certain extent vitamin K1 can also substitute for vitamin K2, but this substitution is limited because our bodies distribute vitamin K1 primarily to the liver and vitamin K2 primarily to other tissues. The specific form of vitamin K2 found in animal fat, moreover, has unique functions that are shared neither by the forms of vitamin K2 found in fermented plant foods nor by the vitamin K1 found in leafy greens.15 As shown in Figure 3, toxicity results when the supply of these vitamins is thrown off balance. When vitamins A, D, and K2 are all available in rich supply, by contrast, as shown in Figure 4, they cooperate to promote growth, to nourish strong bones and teeth, and to prevent the calcification of soft tissues.
When large imbalances between vitamins A and D favor vitamin A, phosphorus accumulates at the expense of calcium, promoting bone loss.16 Vitamin A may also overwhelm the storage capacity of the liver under these conditions, contributing to liver damage. When the imbalance favors vitamin D, calcium accumulates in soft tissues, leading to stones in the kidney and bladder, and calcification of the blood vessels and aortal valves.13 In a growing child, this imbalance would be likely to favor premature calcification of the growth plates, thereby preventing the child from reaching his or her full potential for growth.14 This aberrant pattern of calcification occurs at least in part because the imbalance contributes to the overproduction of vitamin K-dependent proteins in great excess of the capacity for vitamin K2 to activate them.17 These include proteins that direct calcium to our bones and teeth and away from our soft tissues. Since vitamin K2 fails to activate these proteins, the proteins in turn fail to ensure the adequate nourishment of our bones and teeth and fail to protect our soft tissues. One remaining question is whether vitamin K2 protects against vitamin D toxicity just as vitamin A does. This seems likely, but no studies have yet shown it to be true.
One study thus far has demonstrated the interaction between vitamins A and D in humans. In 1941, Irwin G. Spiesman published a trial showing that massive doses of vitamins A and D caused toxicity when either vitamin was provided alone and failed to protect against the common cold. When massive doses of both vitamins were provided together, by contrast, they failed to induce any toxicity and offered powerful protection against the common cold.18 Some authors have argued that a second study published in 2001 showed antagonism between the two vitamins.19 This study, however, did not show a true interaction. Vitamin A decreased blood levels of calcium by 1.0 percent when given alone, and by 1.4 percent when given in combination with the hormone form of vitamin D. The authors did not measure blood levels of phosphorus, and failed to show that vitamin A did anything different in the presence of vitamin D than in its absence.
Recent evidence from experiments performed on isolated cells suggests that vitamins A and D may synergistically suppress the development of autoimmune diseases20 and perhaps even cure diabetes by causing the regeneration of pancreatic stem cells.21 Forming any conclusions from these studies would be premature, however, since we need to follow them up with nutritional studies in humans or live animals.
Altogether, the available evidence supports the rich provision of vitamins A, D, and K2 together by consuming organ meats, animal fats, fermented foods, fatty fish, cod liver oil, and colorful vegetables, while spending plenty of time outdoors. Obtaining a rich supply of these vitamins together allows each of them to carry out its biological functions safely and effectively.
Magnesium : The Universal Metal
Some nutrients play so many roles in the body that literally everything depends on them. One such nutrient is magnesium.22 Magnesium is abundant in many whole grains, nuts, seeds, legumes and vegetables, some fruit, and some seafood. It is less abundant in meat, by contrast, and almost entirely absent from refined grains and sugar (Table 6). Modern diets rich in refined grains and sugar thus provide far less magnesium than traditional diets wherein these “displacing foods of modern commerce” were absent.
Magnesium contributes to more than three hundred specific chemical reactions that occur within our bodies.22 The most basic energy currency of our cells, ATP, exists primarily bound to magnesium. Magnesium is thus essential for every reaction that depends on ATP. Magnesium also activates the enzyme that makes copies of DNA, as well as the enzyme that makes RNA, which is responsible for translating the codes contained within our genes into the production of every protein within our body. Magnesium is thus literally involved in every single process that occurs within the body, making a specific enumeration of all of its interactions impossible to contain even within a large book, far less an article such as this. The well known interaction between magnesium and calcium, however, provides a classic example (Figure 5).
Magnesium deficiency decreases blood levels of calcium in humans and most animals.22 The reasons for this are complex and reflect the universal importance of magnesium rather than a specific interaction between the two minerals. In a healthy individual, parathyroid hormone activates vitamin D to its hormone form, which in turn maintains blood levels of calcium within the appropriate range, in part by helping us absorb calcium from our food.
Magnesium deficiency causes a failure in this system through several mechanisms. Without magnesium, the liver cannot convert vitamin D to its semi-activated storage form, 25-hydroxyvitamin D. When we are deficient in magnesium, we not only produce less parathyroid hormone, but even what we do produce fails to work properly. This resistance to parathyroid hormone appears to result from the failure of at least four different categories of biochemical reactions that are needed to support the hormone. Without properly functioning parathyroid hormone, our kidneys fail to fully activate the storage form of vitamin D to its hormone form, 1,25-dihydroxyvitamin D. On top of all of this, even fully activated vitamin D fails to function properly when we are deficient in magnesium, probably because all of the proteins it controls are at least indirectly dependent on the mineral. This cascade of biochemical failures ultimately depresses calcium absorption, and obtaining sufficient magnesium from food or supplements is the only remedy that will restore calcium levels to normal.
Not only do we fail to absorb enough calcium when we are deficient in magnesium, we also fail to put calcium where it belongs.22 Over 99 percent of the calcium in our body belongs outside of our cells, primarily in our bones and teeth. While only a small amount is found in our blood at any given moment, it is our blood that provides calcium to our bones and teeth where the bulk of it is stored. Only a small portion of calcium belongs inside our soft tissue cells. Our cells keep this small amount in storage vesicles, and release it when needed to stimulate certain functions such as muscular contraction.
Magnesium is needed to utilize the most basic energy currency of our cells, ATP, which is in turn needed to activate the pumps and channels that maintain the proper distribution of calcium and other minerals within our cells. When we are deficient in magnesium, our cells accumulate sodium and lose potassium. The potassium is lost in our urine, while the sodium draws excess calcium into the cell. In the absence of magnesium, our cells are unable to store calcium in the appropriate vesicles. The accumulation of calcium within our cells robs calcium from the blood, which means less calcium is available to our bones and teeth. This total failure of mineral metabolism contributes to excessive excitation of nerves and muscles, disturbances in the rhythm of the heart, a tendency of the blood to clot too much, and poor mineralization of the bones and teeth.
Synergy and Context
The human body is a biological system characterized by astounding complexity. Nutrients often cooperate with one another to produce vibrant health. Quite often when one or more nutrients is missing, others may appear to contribute to disease. Methionine from muscle meats may appear to contribute to disease, for example, when the B vitamins, choline, and glycine found in bones, skin, organ meats, egg yolks, legumes, and leafy greens are absent. Vitamins A and D may each appear to contribute to disease when the other is absent. In the absence of other nutrients such as magnesium, some nutrients such as vitamin D and calcium may simply fail to function at all. The complex biology that makes the human body tick may operate very differently in the context of a diet rich in magnesium than in the context of a diet poor in magnesium.
Nutrient-dense, traditionally balanced diets, however, provide all of these nutrients together so that they synergize with one another to nourish our bodies to health and protect them from harm. Rather than seeking dietary villains from among our most ancient traditional foods to blame for our most recent modern diseases, we should elaborate our understanding of how the many components within successful traditional diets work together to promote radiant and vibrant health.
SIDEBARS
Figure 1: SYNERGY BETWEEN METHIONINE, B VITAMINS, CHOLINE, AND GLYCINE1-3

The figure presents a simplified model of the synergy between methionine, B vitamins, choline, and glycine. See text as well as Figure 2 for a more detailed view.
A. Excess methionine from muscle meats, in the absence of protective nutrients, generates homocysteine, a toxic byproduct that may contribute to vascular disease.
B. With adequate B vitamins and choline, found especially in organ meats and egg yolks, methionine will not generate excess homocysteine, but it may lead to excessive methylation, a breakdown of cellular communication, and depletion of glycine, all of which may contribute to the negative consequences of aging. See text as well as Table 2, Table 4, and Table 5 for additional sources of protective nutrients.
C. Adequate glycine from gelatinous materials such as bones, skin, and other connective tissue, works together with B vitamins and choline to prevent excessive methylation and to ensure adequate conversion of methionine to glutathione. This in turn ensures that methionine will be used for proper cellular communication and as part of the cellular defense against oxidants and environmental toxins.

Animal products have a higher percentage of their total protein as methionine than plant products. Although not shown in the table, they also contain much more protein per unit of weight or volume. The main sources of methionine in the diet, then, are milk, eggs, fish and meat. Although liver and egg yolks provide methionine, they are also rich in nutrients that cooperate with methionine to render it safe and effective (see the main text as well as Table 2 and Table 4). Muscle meats, by contrast, including fish, are rich in methionine but poor in key cooperative nutrients.

Folate is especially abundant in liver and legumes, with moderate amounts in egg yolks, some seeds, some seafood, and some leafy greens, but very little in muscle meats. Folate content tends to be widely variable within a food group, and only a small selection of foods is reported in the table above.
Figures 2A and 2B (below):
METHIONINE METABOLISM AT LOW AND HIGH CONCENTRATIONS OF METHIONINE.1-3
We obtain methionine from most dietary proteins, but primarily from muscle meats. We use it to build our own proteins, but also for two other important processes: methylation and the synthesis of glutathione. Of these, methylation takes priority. Methylation is the addition of one-carbon units to a wide variety of molecules, which aids in the synthesis of many cellular components and in the regulation of gene expression.
When cellular concentrations of methionine are insufficient or just barely sufficient to meet the demand for methylation, the pathways shown in panel A predominate. During each methylation reaction, methionine is converted to homocysteine, which is potentially toxic. Folate and vitamin B12 help recycle homocysteine to regenerate methionine, which allows methylation to continue and prevents homocysteine from accumulating to toxic levels. Although not shown in panel A, niacin, riboflavin and vitamin B6 also assist in this process.
When the supply of methionine exceeds that needed for methylation, the excess is metabolized mainly in the liver and the pathways shown in panel B predominate. Glycine accepts the extra methyl groups, while choline and betaine recycle part of the extra homocysteine. These processes all result in the accumulation of dimethylglycine, part of which is lost in the urine.6 Vitamin B6 and glycine assist in the conversion of part of the extra homocysteine to cysteine and then to glutathione, which is the master antioxidant and detoxifier of the cell, and a key regulator of protein function. When the flux through this latter pathway exceeds the capacity for glutathione synthesis, the excess cysteine is converted to taurine and sulfate. Thus, B vitamins, choline, betaine and glycine all cooperate with methionine to allow optimal methylation and synthesis of glutathione. When methionine is provided in the absence of these partners, methylation and glutathione synthesis fall by the wayside and homocysteine accumulates to potentially toxic levels. It may also be the case that if only glycine is limiting, the capacity to absorb extra methyl groups diminishes and rogue methylations occur.

A. During each methylation (reaction 3), methionine adenosyltransferase uses ATP to convert methionine to S-adenosylmethionine (SAM). Methyltransferases then use SAM to methylate a wide variety of molecules, generating S-adenosylhomocysteine (SAH). S-adenosylhomocysteine hydrolase then allows a small amount of SAH to generate homocysteine, which is potentially toxic. Vitamin B12 and folate assist in the recycling of homocysteine back to methionine. 5-methyltetrahydrofolate (a form of folate, abbreviated as methyl-folate in the figure) methylates vitamin B12 (reaction 1), and methionine synthase then uses B12 to methylate homocysteine and thereby form methionine (reaction 2). Folate is then remethylated through several different pathways not shown in the figure, which depend on niacin, riboflavin, and vitamin B6.

B. When methionine concentrations are high, an alternative set of reactions predominates in the liver. Choline dehydrogenase and betaine aldehyde dehydrogenase convert choline to betaine with the assistance of niacin and oxygen (reaction 1). Betaine-homocysteine methyltransferase uses betaine to convert homocysteine to methionine, generating dimethylglycine (reaction 2), part of which is converted to glycine and part of which is lost in the urine.6 More SAM is generated than is needed for methylation reactions.
With the assistance of vitamin B6, cystathionine b-synthase and cystathionine g-lyase use serine to convert homocysteine to cysteine in two successive steps (reaction 5). Serine is obtained directly in the diet or derived from glycine with the assistance of vitamin B6 (reaction 4). Using glycine, glutamate, and ATP, glutamate cysteine ligase and glutathione synthase convert cysteine to glutathione in two successive steps (reaction 6). Excess cysteine is converted to taurine and sulfate (reaction 7). As a result, glycine, obtained directly from the diet or synthesized from dietary serine with the assistance of vitamin B6 (reaction 4), accepts methyl groups from SAM, generating dimethylglycine (reaction 3), part of which is lost in the urine.6 N-methyltransferases catalyze this reaction in two successive steps.

Glycine makes up only five percent of the amino acids in typical muscle meats, but is much richer in proteins such as collagen and elastin, found in connective tissues like skin and bone. Most of the protein in bone is collagen, which is about one-third glycine, making any dishes made from bone, including soups and sauces, excellent sources of this amino acid.



Figure 3: IMBALANCES BETWEEN VITAMINS A AND D LEAD TO TOXICITY13,16,17
When a severe imbalance between vitamins A and D favors vitamin A, phosphorus accumulates at the expense of calcium and bone loss ensues. When such an imbalance favors vitamin D, the production of vitamin K-dependent proteins greatly exceeds the capacity of vitamin K to activate them. This results in defective proteins, which in turn fail to direct calcium away from soft tissues and into bones and teeth. This results in soft tissue calcification, including the formation of kidney and bladder stones, and the calcification of blood vessels and aortal valves.

SYNERGY BETWEEN VITAMINS A, D, and K213,14,17
When vitamins A and D are available in an appropriate balance, cells produce a healthy amount of vitamin K-dependent proteins. When vitamin K2 is available in rich supply to activate them, these proteins protect against soft tissue calcification and direct calcium to where it belongs: in the bones and teeth. During growth, these proteins protect the growth plates from premature calcification, ensuring that a child will reach his or her full growth potential.

Table 6. Magnesium Content of Selected Foods23
Magnesium is especially abundant in many seeds, and also found abundantly in many whole grains, nuts, and vegetables. Some fruits and types of seafood are also good sources. Meat, however, even liver, is low in magnesium. In contrast to whole grains, refined grains contain almost no magnesium. Like refined grains, refined sugar also contains almost no magnesium.

Figure 5: Contribution of Magnesium to Calcium Metabolism 22
Magnesium (Mg2+) is necessary for virtually every function in the body. As a result, proper calcium metabolism breaks down in the absence of sufficient magnesium. Magnesium helps convert vitamin D to the semi-activated storage form, 25-hydroxyvitamin D, abbreviated in the figure as 25(OH)D. It contributes both to the production of parathyroid hormone (PTH) and to its conversion of 25(OH)D to the fully activated hormone form of vitamin D, 1,25-dihydroxyvitamin D or 1,25(OH)2D. Magnesium helps 1,25(OH)2D stimulate calcium absorption, and assists the variety of pumps and channels that help distribute calcium properly into the bones and teeth, blood, and storage vesicles where it belongs. In the absence of sufficient magnesium, we fail to absorb enough calcium from our food. The calcium we do absorb accumulates within our cells rather than in our blood, bones, and teeth, where it belongs. Our cells, moreover, fail to sequester it in storage vesicles. These changes as well as other failures of mineral metabolism that occur during magnesium deficiency contribute to excessive excitation of nerves and muscles, blood coagulation, and poor mineralization of bones and teeth.
REFERENCES
1. Melendez-Hevia E, De Paz-Lugo P, Cornish-Bowden A & Cardenas ML. A weak link in metabolism: the metabolic capacity for glycine biosynthesis does not satisfy the need for collagen synthesis. J Biosci. 2009;34(6): 853-872.
2. Martinov MV, Vitvitsky VM, Banerjee R & Ataullakhanov FI. The logic of the hepatic methionine metabolic cycle. Biochim Biophys Acta. 2010;1804(1): 89-96.
3. Carmel R. Folic Acid. In Modern Nutrition in Health and Disease (eds M.E. Shils et al.) (Lippincott Williams & Wilkins, 2006).
4. Stipanuk MH. Homocysteine, Cysteine, and Taurine. In Modern Nutrition in Health and Disease (eds M.E. Shills, et al.) 545-562 (Lippincott Williams & Wilkins, 2006).
5. Bleie O et al. Changes in basal and postmethionine load concentrations of total homocysteine and cystathionine after B vitamin intervention. Am J Clin Nutr. 2004;80(3): 641-648.
6. Atkinson W, Elmslie J, Lever M, Chambers ST & George PM. Dietary and supplementary betaine: acute effects on plasma betaine and homocysteine concentrations under standard and postmethionine load conditions in healthy male subjects. Am J Clin Nutr. 2008;87(3): 577-585.
7. Olthof MR, Brink EJ, Katan MB & Verhoef P. Choline supplemented as phosphatidylcholine decreases fasting and postmethionine-loading plasma homocysteine concentrations in healthy men. Am J Clin Nutr. 2005;82(1): 111-117.
8. Brind J, et al. Dietary glycine supplementation mimics lifespan extension by dietary methionine restriction in Fisher 344 rats. FASEB J. 2011;25(528.522.
9. McCarty MF, Barroso-Aranda J & Contreras F. The low-methionine content of vegan diets may make methionine restriction feasible as a life extension strategy. Med Hypotheses. 2009;72(2): 125-128.
10. Meakins TS, Persaud C & Jackson AA. Dietary supplementation with L-methionine impairs the utilization of urea-nitrogen and increases 5-L-oxoprolinuria in normal women consuming a low protein diet. J Nutr. 1998;128(4): 720-727.
11. Masterjohn C. Vegetarianism and Nutrient Deficiencies. Wise Traditions. 2008;9(1).
12. Tang G, Qin J, Dolnikowski GG & Russell RM. Short-term (intestinal) and long-term (postintestinal) conversion of beta-carotene to retinol in adults as assessed by a stableisotope reference method. Am J Clin Nutr. 2003;78(2): 259-266.
13. Masterjohn C. From Seafood to Sunshine: A New Understanding of Vitamin D Safety. Wise Traditions. 2006;7(2).
14. Masterjohn C. On the Trail of the Elusive X Factor: A Sixty-Two-Year-Old Mystery Finally Solved. Wise Traditions. 2007;8(1).
15. Ichikawa T, Horie-Inoue K, Ikeda K, Blumberg B & Inoue S. Vitamin K2 induces phosphorylation of protein kinase A and expression of novel target genes in osteoblastic cells. J Mol Endocrinol. 2007;39(239-247.
16. Masterjohn C. Vitamin A on Trial: Does Vitamin A Cause Osteoporosis? Wise Traditions. 2006;7(1).
17. Fu X et al. 9-Cis retinoic acid reduces 1alpha,25-dihydroxycholecalciferol-induced renal calcification by altering vitamin K-dependent gamma-carboxylation of matrix gammacarboxyglutamic acid protein in A/J male mice. J Nutr. 2008;138(12): 2337-2341.
18. Spiesman IG. Massive Doses of Vitamins A and D in the Prevention of the Common Cold. Arch Otolaryngol Head Neck Surg. 1941;34(4): 787-791.
19. Johansson S & Melhus H. Vitamin A antagonizes calcium response to vitamin D in man. J Bone Miner Res. 2001;16(10): 1899-1905.
20. Ikeda U, et al. 1alpha,25-Dihydroxyvitamin D3 and all-trans retinoic acid synergistically inhibit the differentiation and expansion of Th17 cells. Immunol Lett. 2010;134(1): 7-16.
21. Ng KY, Ma MT, Leung KK & Leung PS. Vitamin D and vitamin A receptor expression and the proliferative effects of ligand activation of these receptors on the development of pancreatic progenitor cells derived from human fetal pancreas. Stem Cell Rev. 2011;7(1): 53-63.
22. Rude RK & Shils ME. Magnesium. In Modern Nutrition in Health and Disease (eds M.E. Shills et al.) 223-247 (2006).
23. SelfNutritionData: know what you eat., <http://nutritiondata.self.com>, accessed August 27, 2012.
24. Howe JC, Williams JR & Holden JM. USDA Database for the Choline Content of Common Foods. U.S. Department of Agriculture Agricultural Nutrient Data Laboratory. 2004.
This article appeared in Wise Traditions in Food, Farming and the Healing Arts, the quarterly magazine of the Weston A. Price Foundation, Fall 2012.
🖨️ Print post

Numbers?
Nice article (except for one typo: in the second bullet point of the summary you probably mean “leafy greens” instead of “leafy grains”)!
However, you don’t seem to put any numbers on how to achieve balance. For instance, I just typed in what I ate today at an online calculator, and it says I got 470 mcg folate, 1.9 g methionine and 4.2 g glycine (all from food). Is that balanced? It doesn’t tell me how much choline I had, but I had two eggs. Also, can we even trust the numbers at online nutrition databases?
What number should we aim for? Do you generally agree with the suggested nutrient levels that the Jaminets have in their book (e.g., the new Scribner edition)?
As usual
Great article! 🙂
So, is fish gelatin a good source of glycine for fishatarians? How much should we be aiming for?
sources of magnesium
the values listed in table 6 for tomatoes, bell peppers, bananas, and peaches are calculated from the dry mass. as people don’t normally eat large quantities of dried tomatoes and bell peppers, this is somewhat misleading. frankly, i’m surprised there was no mention of whelk or snail, given the bias toward ancestral diets.
phosphate fertilizers used in nut and grain production increase the concentration of phytic acid in the bran, which limits the amount of magnesium we can absorb from nuts and grains. simply eliminating sugar and refined grains will not correct a magnesium deficiency, it has to be supplemented. don’t believe me? do the math on your own diet and see if you actually meet the RDA for magnesium.
Replies to Fenn, James, Andrea, and Ulrik
Fenn,
I didn’t realize the numbers were for dry matter. That is surprising. Could you direct me to your source for that information? Thank you.
The tables were not meant to be comprehensive. The article was not a general article about magnesium, so naturally much that could be said about magnesium was left out. Surely whatever improvements could be made to a diet that excludes white flour and sugar and isn’t based entirely off meat don’t negate the dramatic impact eating a wide variety of unrefined plant foods could have on magnesium intake.
James,
It should be. We really don’t know the amount that should be consumed, but as I indicated in the article it could be ten grams of glycine, which would be found in about an ounce of bone.
Andrea, thanks!
Ulrik, thanks for pointing out the typo. I’ll have it fixed. I did give some numbers, but there isn’t anything definitive to say about them. One group suggested we might need 10 grams of glycine per day, in which case you’d be a bit short. Defining needs for folate, choline, etc, is incredibly difficult because genetics impact the folate requirement and folate and choline each reduce the need for the other. There isn’t enough information to quantify this interaction. Paul sent me a copy of the hardcover version of the new book but I haven’t had a chance to read it yet. I’ll try to do that and review it.
Thanks everyone!
Chris
The PDF of this otherwise wonderful article, is unreadable.
Consuming fat
Chris, the article says to consume animal fat, what is the recommended way to do that?
Won’t consumption of betaine-glycine increase glycine levels? When betaine loses its methyl groups, the end result is glycine. Also, the intermediate product, sarcosine (i.e. methylglycine), inhibits the type 1 glycine transporter.
Wow
:);D Aboslutly beautifuly presented!
Another great article, Chris!
Just learned I have compound heterozygous MTHFR mutations (aka: both 677 and 1298…thanks, Mom & Dad!), and am trying to figure out if and how I need to supplement. I have been eating a strict very-low carb variant of a WAPF diet for 2.5 years, supplementing with desiccated liver (1.25 oz / day), FCLO (2 tsp / day), HVBO (1 tsp / day), wild salmon roe (1 tsp / day), exp-pressed coconut oil (2 tbsp / day), pastured eggs (2 raw yolks / day), and raw grass-fed milk (12-18 oz / day).
My MD prescribed Foltx, but I haven’t taken it since I’m trying to get pregnant and its effects are unknown in pregnancy. Also, I had my b-6, b-9, and b-12 plasma levels drawn: b-9 is high (19.9 ng/mL), and both b-6 and b-12 are mid-normal (12.6 ng/mL & 501 pg/mL respectively). Where would you recommend I self-educate about what to take and what/when to re-test? Thx!
Check out this web site regarding MTHFR mutations. I have found it very helpful.
Colleen, what is the website?
Carol, check out Dr. Ben Lynch at mthfr.net http://mthfr.net/what-is-mthfr/2011/11/04/ Also the Phoenix Rising site at http://phoenixrising.me/ might be helpful, it’s focus is not the MTHFR defects but CFS/ME.
I was wondering if you understand how difficult it is today for us to get adequate amounts of magnesium through our food supply due to depleted soils and chemical fertilizers, not to mention refinement of grains and taking prescriptions. And because of this, an underlying magnesium deficiency is responsible in part for most of illness today. This article is a few years old, so maybe you’ve changed your ideas on this, but you don’t mention these problems in the article. If so, I’d love to hear your take on it. Thanks!
Great article, not sure how I missed it.
Figure 1 in particular (showing different outcomes of methionine metabolism) explains a lot.
Thank you Chris!
I’m convinced that the “standard American diet”(SAD) drains Mg from the bodies of most individuals on that diet. WAPFers may not be doing SAD however most of our soils are “sad”. I think we need to know what our farmers and growers have to do to fully remineralize their soils and then see that they actually do that. If we can’t assure the inclusion of minerals at the source as well as in the foods (and at a level comparable to what Weston Price found) then the next option is probably supplementation.
For supplementation I suggest a unique product[1] that was developed by Dr. Carolyn Dean, author of The Magnesium Miracle.
Have been blogging on Magnesium[2] over the past fifteen months. The more I read on this the more amazed I become!
Read: Free e-books by Dr. Dean:
https://drcarolyndeanlive.com/category/books/
[1] https://www.rnareset.com/pages/remag-highly-absorbed-magnesium
[2] https://www.curezone.org/blogs/fm.asp?i=2389650
It’s been a long time. I know. But you can enjoy other people.
“… we need to know what our farmers and growers need to do to completely remineralize their soils … it’s hard to get magnesium in the diet …”
Yes. Plants look for it, transport it and we eat it already missing. But it does not return to the soil almost never, making the cycle. Mg is very soluble. It is very washable from the soil. By watering or raining, it always ends up at sea.
Fertilization takes about 2% Magnesium Nitrate, but neither can the vegetative activity of an annual plant and the rest goes to the sea always. Small parts contained or blocked in the soil clay are very little in general.
All to understand that old soils (not those of lava from rich volcanoes. Japan, southern Italy, etc.) are almost always poor. So if it’s gone, that’s left, that’s where we have to get it – overboard. And it’s easier than it sounds.
If we take sea salt (Sodium Chloride) from the sea, we also take Magnesium Chloride (Mg2 Cl) at the same time. And, buy 100 g packaging will give for 4 to 8 months of supplement. Namely, in the supplement of 320 to 420 mg / day / adult, over 45 to 50 years, not obese, not consuming diuretics, ruled with alcohol, (others need a little more) be obtained as follows: 6 gr = 6000 mgr. Pour 1.5 (coffee spoon) into 1 liter of water has about 10 small cups. When you take a cup of this water you have 6000:10 = 600 mgr / cup, but as about 30% is lost in the gut, then there is 70% of 600 mg left or about 420 mg / day ….
I take it every day divided into 2 sparse parts and it has been many years with much satisfaction and health
Both Glycine and Serine deficiencies are very common today due to herbicides (Glyphosate) pesticides (Gluphosanate) and solvents. They also deplete 4 other amino acids. In light of this discovery (by my nutrition partner), we need to both supplement them, and re-examine all causes behind diseases, disorders, and dysfunctions. For example, anxiety and depression, addictions, Type II Diabetes, transgenderism, and many brain problems are deeply rooted in l-serine deficiency.
Thank you for this info. I think you might be on to something.
Nice article Chris, liked the way you simplified the interactions between Mg & vitamin D (Fig 5). We also recently focused on similar areas (https://jaoa.org/article.aspx?articleid=2673882). We believe: without optimal Mg balance, taking vitamin D supplements might not be useful.
All the info in this article is highly informative. But, by God, we were not designed to eat according to complicated scientific formulas! As I see it, you could apply all, and I mean all, of Chris’ information to your diet – and still suffer this or that. And as virtually all health researchers do, they will find an excuse when they eventually develop some problem.
Basically, we are being regarded as machines; you know, stuff in the right fuel and oil and there you go, you are “healthy”. I am grateful for the technical material supplied here but there are human physical problems which are going to pop up in any case. There’s way more to know than just following a nutrient-rich diet.
There are reasons why every race, every culture, every ethnic and every tribal group in all of history developed systems of medicine and that is because they knew (though not consciously) that eating the right food doesn’t always cut the mustard. “Man proposes; God disposes.” Thanks for reading.